НОВИНКИ

Функциональная грамотность. Развитие креативного мышления и глобальной компетенции. 4 класс.
Автор-составитель: Попова Е. Ю.
Страниц: 80 с.

Функциональная грамотность. Развитие креативного мышления и глобальной компетенции. 3 класс.
Автор-составитель: Дейнека Н. Н.
Страниц: 80 с.

Функциональная грамотность. Развитие креативного мышления и глобальной компетенции. 2 класс.
Автор-составитель: Дейнека Н. Н.
Страниц: 80 с.






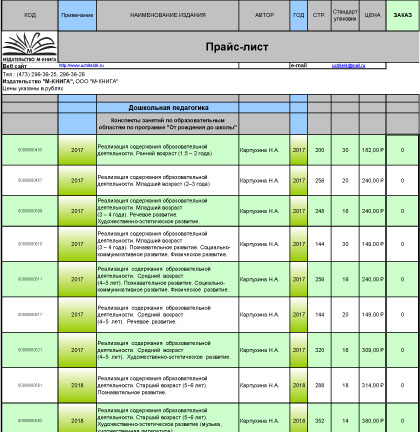
КАТАЛОГ
ИЗДАТЕЛЬСТВО «М-КНИГА»
Большой опыт
Работаем более 15 лет
Высокое качество
Материалы лучших авторов
Большой выбор
Все издания в наличии
Разумные цены
Цены ниже чем у конкурентов
Издательство «М-КНИГА» – издательско-торговая компания. Наша компания занимается изданием и реализацией учебно-методической литературы как собственных пособий, так и пособий других издательств. Цель нашей компании – издание качественной учебно-методической литературы для дошкольного и школьного образования и воспитания, направленную на практическую помощь в работе учителей общеобразовательных школ, методистов, воспитателей дошкольных образовательных учреждений. Наши издания всегда востребованы на рынке учебно-методической литературы. Авторы, сотрудничающие с нашим издательством — это педагоги-практики высокого профессионального уровня, с большим педагогическим опытом и стажем работы в образовательных учреждениях, лауреаты и дипломанты Всероссийских профессиональных конкурсов.
Помимо литературы собственного издания мы реализуем пособия других издательств. В нашем магазине всегда имеется широкий ассортимент учебно-методических, наглядных, дидактических пособий, рабочих тетрадей для образовательных учреждений.
Учебная литература нашего издательства направлена на практическую помощь в работе учителей, методистов и воспитателей. Наши издания всегда востребованы на рынке учебно-методической литературы. Авторы, сотрудничающие с нашим издательством — это педагоги-практики высокого профессионального уровня, с большим педагогическим опытом и стажем работы в образовательных учреждениях, лауреаты и дипломанты Всероссийских профессиональных конкурсов.
Мы более 15 лет на рынке учебно-методической литературы! За это время издательство зарекомендовало себя как стабильная организация и надежный партнер, всегда выполняющий свои обязательства перед поставщиками и покупателями.